Alteration of Serum Reproductive Hormones and the Risk of Preeclampsia in Pregnancy: A Longitudinal Study in Gravid African Women
Ganiyu O. Adeosun1, Mabel A. Charles Davies2, Omobola A.Ogundahunsi3, Jaye Ogunlewe3, Ibrahim S. Bello4 and Tonia Chidinma Onyeneke5
1Department of Chemical Pathology, Obafemi Awolowo University Teaching Hospital Ile-Ife, Nigeria
2Department of Chemical Pathology & Immunology, University College Hospital Ibadan, Nigeria
3Department of Chemical Pathology & Immunology, Olabisi Onabanjo University Ago-Iwoye, Nigeria
4Department of Family Medicine Obafemi Awolowo University Teaching Hospital Ile-Ife, Nigeria
5Department of Medical Laboratory Science, Andrews University, Michigan, MI 49104, USA
*Address for Correspondence: Ganiyu Oyebola. Adeosun, Department of Chemical Pathology, Obafemi Awolowo University Teaching Hospital, PMB5538 Ile-Ife, Nigeria, Tel: +234-803-714-8125; E-mail: [email protected]
Submitted: 15 October 2019; Approved: 17 December 2019; Published: 19 December 2019
Citation this article: Adeosun GO, Charles Davies MA, Ogundahunsi OA, Ogunlewe J, Bello IS, et al. Alteration of Serum Reproductive Hormones and the Risk of Preeclampsia in Pregnancy: A Longitudinal Study in Gravid African Women. Int J Reprod Med Gynecol. 2019;5(2): 059-066.
Copyright: © 2019 Adeosun GO, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited
Keywords: Preeclampsia; Predictors; β-hCG; Oestriol
Download Fulltext PDF
Introduction: Preeclampsia is a medical emergency implicated in maternal and foetal morbidity and mortality. Research directed at finding the aetiologies of pre-eclampsia is inconclusive because most studies are either cross-sectional or semi -longitudinal in design with very limited potentials to determine precisely the age of pregnancy to suspect the development of preeclampsia with an absolute biomarker.
Objectives and Aim: The main aim of this study was to establish the gestational age of pregnancy at which serum hormones alterations became significant to suspect the development of preeclampsia in pregnancy.
Study design: The study was longitudinal undertaken at the antenatal clinics of a teaching hospital and conducted in two phases. The cross-sectional study was conducted among 79 pre-eclamptic pregnancy and 80 normotensive healthy pregnant women monitored from 3rd trimester to 3 days post-partum., while the longitudinal study involved 10 preeclamptics and 20 normotensive healthy pregnant women monitored from the first trimester of pregnancy to six weeks post-delivery.
Methods: Venous blood samples obtained from the participants at each point of contact during the study period were placed into none anti-coagulated specimen bottles. The serum harvested after clot retraction was assayed for β human chorionic gonadotrophic hormones, oestrogen, progesterone, and prolactin by Enzyme Immunosorbent Assay Method (EIA) described by immunometric, Atlanta SW6 6TU (UK) LTD
Result: β-hCG (Odd Ratio = 0.995, confidence interval = 0.998-0.999, p < 004) and oestriol (Odd Ratio = 1.056, confidence interval = 1.011-1.103 =, p < 0.01) only were significantly elevated in the preeclamptic pregnancies compared with the controls. The alteration predicted the development of preeclampsia at first and third trimesters of pregnancy respectively.
Conclusion: Early pregnancy significant alterations of serum level of β-hCG and oestriol and is associated with the risk of development of preeclampsia in gravid women.
Introduction
Preeclampsia (PE), Eclampsia (EP) and other hypertensive disorders in Pregnancy represent the highest portion of maternal mortality and perinatal morbidity in Nigeria as in most developing countries around the world [1]. PE was defined by [2] as the increase in arterial pressure levels after 20 weeks of gestation (pressure level ≥ 140×90 mmHg in two measurements with an interval of 6 hours followed by the presence of proteinuria (1+ or more in the measurement of proteinuria with test strips or 24- hour proteinuria > 0.3 g/24 hours). Studies show that the incidence of preeclampsia is about 4-8% of pregnancies in developing countries [3] but in southern Nigeria, the prevalence is put at 5.6- 7.6% [4,5] and is 40% in northern Nigeria [6]. Untreated PE progresses to eclampsia which portends substantial risks for the fetus and the mother [7,8]. The causes of PE are not yet completely understood, however, accumulated evidences implicated abnormal placenta as the site of origin of the disease [9-11].
Materials and Methods
Study design
The study was longitudinally conducted in two phases; semi and full longitudinal phases. The study and control subjects were followed up from the early pre-delivery pregnancy period to six weeks postpartum through antenatal and post-natal clinic appointments.
Study setting
The study was conducted between January 2018- March 2019. The participants were recruited from the antenatal clinics and wards of a Tertiary Teaching Hospital in Ile-Ife- Southwest Nigeria. Whole blood was collected into a plain bottle at each trimester of the pregnancy during the antenatal and postnatal clinic appointments. Serum was harvested after clot retraction and preserved for the β hCG, Oestriol, Progesterone and Prolactin assays using standard enzyme immunoassay method described by Sachidhanandam, et al. [14].
Baseline values
Baseline values of biophysical clinical and biochemical indices for statistical analyses were taken at a mean gestation age of 10.9 ± 0.3 weeks for the full longitudinal phase and at mean gestation age 35.7 ± 0.0.4 weeks for the semi-longitudinal phase. The biophysical and clinical data were obtained from the patients’ case notes while biochemical data were results from the assay of the reproductive hormones.
Diagnosis of Pre-Eclampsia
PE was diagnosed according to the criteria defined by the National high blood pressure education program working group on high blood pressure in pregnancy [15].
Ethical consideration
Recruitment of participants for the 2- year study started after study protocol was approved by the local Ethics and Research committee of a tertiary hospital (Obafemi Awolowo University Teaching Hospitals Complex Ile-Ife.
Statistical analysis
The data from the study were analyzed using SPSS version 21.0. Continuous variables were presented as the mean and standard error of the mean. While categorical variables were presented as percentages. Differences between groups were compared using a two-tail independent t-test of significance at 95% confidence limit, p < 0.05 was considered significant for the variables. Logistic regression model to predict a dichotomous outcome from one or more predictor variables and the significance of each predictor in the model was tested while all other predictors were held constant. The effect of size for the individual predictor was expressed as an Odds Ratio (OR).
The study experienced loss of patients at both phases of the study but this was curbed by educating the patients further about the study and reaffirmation of consent participation. Data of participants who declined consent before and during the study period were completely deleted from the statistical analysis of the results obtained from this research. Therefore the analyzed data excluded incomplete information or data of participants who were not able to fulfill the study criteria as described in the study design and eligibility conditions.
Results
The eligibility criteria included normotensive and non-proteinuric antenatal cases registered before 13 weeks of gestation that voluntarily gave consent to participate and completed the study.
Exclusion criteria: This comprises hypertensive and proteinuric pregnancy before or at 13 weeks of gestation and recruited subjects who declined consent during the study period.
Sources and methods of selection of participants: The study was undertaken at the Obafemi Awolowo University Teaching Hospital Ile -Ife southwest Nigeria and was conducted in two phases; phases one and two.
Phase one: semi-longitudinal study: Three hundred and fifty pregnant women in the third trimester of pregnancy (28-40 weeks gestation (15) with a mean age of 30 ± 0.7 years were selected for the study through the antenatal clinics of the hospital after obtaining informed consent. From the 350 pregnant women, 79 of them only at the mean gestation age of 35.7 ± 0.0.4 weeks had features of pre-eclampsia [15] and served as the study group. Eighty out of the remaining 271 pregnant women who were physically healthy and normotensive after the screening using the criteria [15] of National High Blood Pressure education program working group on high blood pressure in pregnancy were used as controls. The study and control group were followed up from the point of selection( mean gestation age of 35.7 ± 0.0.4-38.4 ± 0.2 weeks respectively up to 3 days postpartum through their antenatal and postnatal clinic day appointments The remaining 191 subjects did not meet the study criteria or could not complete the study were exempted from the study.
Phase two: This was the longitudinal phase of the study which was a follow up of phase one study to detect the onset of the abnormalities observed in phase one of this study. One hundred physically healthy normotensive pregnant women with mean gestation age between 8-13 weeks and non-proteinuric were recruited from the antenatal clinics of the Tertiary Teaching Hospital after obtaining their informed consent. They were screened for early pregnancy abnormalities using the criteria [15] described above and were followed up from mean gestation age 10.9 ± 0 weeks to 6 weeks post-delivery through their antenatal and postnatal clinic day appointments. During the study period, thirty (30) participants met the study criteria while ten (10) out of these 30 participants developed pre-eclampsia and were grouped as cases of interest while twenty (20) who remained normotensive and non- proteinuric throughout the study served as controls for the study. The remaining seventy (70) subjects were excluded because they did not meet the study criteria. Blood samples obtained from the participants at each point of contact in the study period were assayed for the reproductive hormones using standard enzyme immunoassay method.
Sample size calculation: The sample size was calculated using the formula described by (Rendon-Macias, et al. 2017). [16].
Sample size = Pq (z) 2 /e2 [16].
Where: Z = Standard deviation, e = Margin of error, p = prevalence of preeclampsia in the study area.
Phase one
Three hundred and fifty subjects participated in this phase of the study, from this figure seventy-nine were preeclamptic and met the eligibility criteria and as well gave consent to participate in the study. Eighty subjects had sustained normotensive blood pressure throughout the study period gave consent and served as control. The study and control groups were followed up from the point of recruitment (35-38 weeks) up to 3 days postpartum. The remaining one hundred and ninety-one subjects were excluded due to the decline of consent sequel to doubt of completing antenatal care at the study centre, and some having other pregnancy complications like diabetes, renal disease, hypertension and anaemia hence their data were excluded.
Table 1 shows the demographic characteristics of pre-eclamptic and normotensive pregnancies. The incidence of pre-eclampsia was higher (p < 0.05) within the age bracket of 25-34. Gestational age at delivery among the preeclamptics was significantly lower compared with the controls (p < 0.05). Delivery by Caesarian Section in preeclampsia was 57 (81.4%) and 17 (24.3%) progressed to eclampsia. Two maternal 2 (2.9%) and nine neonatal deaths 9 (12.9%) b were recorded among the preeclamptics. Baseline and postpartum values BMI and blood pressure of cases and controls are presented (Table 2). Systolic and diastolic blood pressures were higher (p < 0.001) among the preeclamptics when compared with normotensive pregnancy (Table 2). Reproductive hormones level at pre and postpartum were compared between preeclampsia and normotensive pregnancy (Table 3). β- hCG and Oestriol only at prepartum were significantly lower and higher respectively (p < 0.001) in preeclampsia when compared with the normotensive controls. Comparison of baseline clinical and biochemical characters of mild and severe preeclampsia are presented in table 4. Systolic and diastolic blood pressures in severe preeclampsia were significantly elevated (p < 0.001) while the differences in the levels of the reproductive hormones between the two groups were not significant (p > 0.05)
Phase two: Table 5 shows the comparative analysis of anthropometric and clinical indices in both study and control subjects from the first trimester to six weeks postpartum. There was no significant difference in the anthropometric indices (p > 0.05) between study and control groups throughout gestation and postpartum. Mean systolic and diastolic blood pressures in preeclamptic patients were significantly elevated (p < 0.000) when compared with the normotensive controls except in the 1st trimester. The alterations in the mean values of micro-albumin in preeclamptic patients throughout the study periods have a similar pattern with that of the blood pressures (p < 0.001).
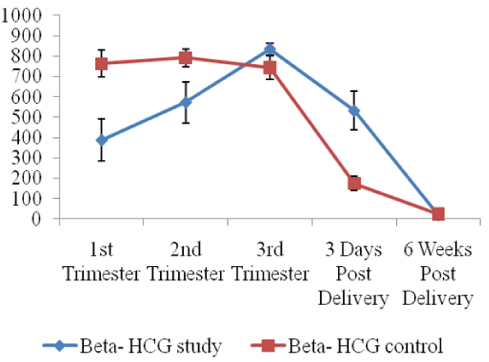
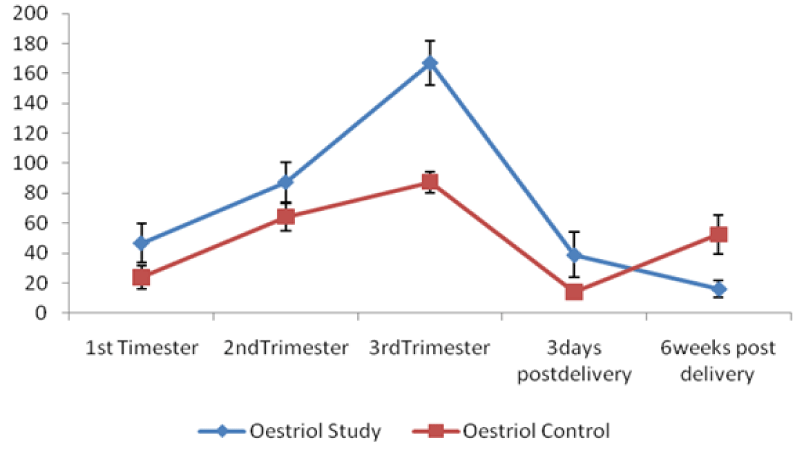
Steady and progressive changes in the mean values of reproductive hormones among the pre-eclamptic group studied from 1st trimester to 6weeks postpartum were evaluated and compared with the normotensive control pregnant women (Table 6, figures 1,2). The mean values of β human chorionic gonadotropin, prolactin, progesterone, oestriol increased steadily and significantly from the first trimester, peaking in the third trimester and declined sharply soon after birth to almost pre-pregnancy level at six weeks post-delivery. (p < 0.05, p < 0.001). β hCG levels in the study group at 1st and 2nd trimesters were significantly lower. (p < 0.05, p < 0.001) relative with the controls. Oestriol concentration at the 3rd trimester only in preeclampsia was observed to be significantly higher (p < 0.001) when compared to the control. The differences in progesterone and prolactin levels throughout the study period between the controls and the preeclamptics were not significant (p > 0.05). The result of the logistic regression analysis to validate the predictive potentials of the reproductive hormones throughout the study period is presented in Table 7. β hCG (OR = 0.995, CI = 0.998-0.999, p < 0.004) and oestriol (OR = 1.056, CI = 1.011-1.103, p < 0.013) have significant predictive potentials of pre-eclampsia at first and third trimesters respectively. (p < 0.004, p < 0.013).
Discussion
Pre-eclampsia and eclampsia which present mostly late in pregnancy are medical emergencies in Nigeria and in most of the world [17]. They are one of the major causes of maternal -perinatal morbidity and mortality worldwide [18] yet no single or combination of pregnancy indices have been found to reliably suspect and prevent this disease [18-20]. Identification of markers of pre-eclampsia is very desirable for ease of early intervention, close monitoring, and prompt diagnosis to reduce the negative consequences of preeclampsia on the nation’s women population.
Phase one of this present study recorded low mean gestation age at term, delivery by caesarian section, neonatal and maternal death were recorded more often among the preeclamptic when compared with the normotensive controls (Table 1). These observations suggest that preeclampsia is associated with adverse pregnancy complications as observed in this study and comparable with the reports of previous studies [21,22]. Preeclampsia can degenerate to mild or severe complications if poorly managed or when intervention is delayed [23]. We compared clinical and biochemical parameters of the mild and server preeclamptics (Table 4). Blood pressure values of severe preeclamptics were significantly elevated compared to the mild form of the disease while serum β -hCG level only in severe preeclampsia was higher but not statistically significant. This observation is at variance with the report of Kanika, et al. [24] who reported elevated serum β-hCG among severe preeclamptics.
Our observations in phase one of the present study show that β-hCG and oestriol were significantly reduced (p < 0.001) and elevated (p < 0.05) respectively in the 3rd trimester among the preeclamptics population when compared with normotensive controls (Table 3). Pre-eclampsia is reported be a multi-organ disease that induces tissue and organ defects to the liver, kidney, placenta, and heart [24,25] The effect of the damaged organs particularly the liver and the placenta arising as a secondary complication of pre-eclampsia and the associated metabolic abnormalities may be the major contributory factors to the significant alterations of β-hCG and oestriol concentrations observed in the pre-eclamptic subjects. We observed that β-hCG and oestriol alteration implicated as possible risk factors pre-eclampsia in phase one of this study was inconclusive because of the gestation age of pregnancy at which β-hCG and oestriol became significant to precipitate pre-eclampsia could not be determined. The outcome of the longitudinal assessment of the reproductive hormones in the pregnant subjects in phase two of this study was designed to fill this missing gap.
Normal endocrine physiological processes in normotensive pregnancy are associated with rising β -hCG rapidly soon after fertilization and implantation and continued throughout the first trimester of pregnancy and peaks at 6.8-10 weeks of pregnancy [26,27]. In the longitudinal phase of this study, we observed significant reduction (p < 0.001), of β hCG in the 1st and 2nd trimesters among the preeclamptics when compared with the control group (Table 6, figure 1). The causes of low β-hCG in the preeclamptics observed at 10.9 weeks of pregnancy that precipitated preeclampsia in our study may be attributed hyperemesis gravidarum in the second trimester induced by high levels of hCG as a compensatory mechanism to low β-hCG early in pregnancy [28-31]. The multi-organ disease nature of preeclampsia that induces disorders of placental dysfunction and abnormal placentation has also been implicated in the cause of significant alteration of β-hCG in complicated pregnancy [32,33].
We also analyzed the changes in β hCG concentration among the preeclamptics only within the study duration. Result revealed a significant progressive increase in β hCG level throughout gestation (p < 0.001). This observation aligned with Kanika, et al [24] and Camilla, et al. [34] findings who reported a consensus that the placenta remains the main source of excess hCG production in patients with pre-eclampsia and other associated pregnancy disorders. Similarly our observation of significant (p < 0.001) progressive rise in β-hCG concentration from 10.9th week of gestation within the preeclamptics in a longitudinal study is an improvement on earlier reports that observed the rise in β-hCG concentration after 15 weeks of gestation which precipitated the development of preeclampsia [31,35,36].
Progressive increase in oestriol levels throughout gestation is a characteristic of pregnancy. In this study, we observed higher values of oestriol concentration among the pre-eclampsia in the 34.5th week of gestation compared with the controls (p < 0.001). Our observation is similar to the reports of Tongprasertet, al. [38], Settiyanan, et al. [39] and Singh, et al. [39] who established the usefulness of oestriol assay in the 3rd trimester in the diagnosis of pregnancies complicated by post maturity, moderate and severe pre-eclampsia, intrauterine growth retardation, diabetes mellitus, chronic hypertension, intrauterine growth retardation. The pathological alterations of β hCG and oestriol in preeclampsia suggest that these hormones are associated with pregnancy-specific diseases and might serve as an effective biomarker for predicting and diagnosing preeclampsia. Furthermore, the result of the logistic regression analysis conducted to evaluate the potentials of reproductive hormones to predict future development of pre-eclampsia in pregnant women strongly suggested that β hCG and oestriol might serve as an effective biomarker for suspecting and diagnosing pre-eclampsia (Table 6). β hCG (Odd Ratio of 0.995, CI of 0.998 - 0.999 , at p < 004) predicted the development of preeclampsia as early as at 10.9th week of gestation, while oestriol (Odd ratio of 1.056, CI of 1.011-1.103 at p < 0.01) predicted the development of pre-eclampsia at 34.5th week of gestation. The prediction of pre-eclampsia at 10.9th week of gestation by β hCG in this study is an improvement on the previous reports by Gurmandeep, et al. and Goetzinger, et al. [31,40] who have earlier reported that maternal low serum β hCG level at 13-20 weeks and 15 weeks gestation respectively is a predictor of pre-eclampsia.
Conclusion
This study concluded that alterations of serum β-hCG and oestriol in pregnancy is pathologic and is associated with the risk of developing preeclampsia. β- hCG and oestriol may constitute a panel of biomarkers for the disease. Early alteration of serum β-hCG observed in the first trimester preceded the onset of preeclampsia most often detected in the third trimester hence pregnancy at the risk of preeclampsia can be identified at early gestation for quick intervention and management.
Acknowledgment
The authors are grateful for the support and the cooperation of the Ethical and Research Committee, the Consultants Obstetrics and Gynaecologist and Nurses of the centre used for this study for granting us access to the patients. We appreciate the professional staff in the laboratory and as well as the cooperation of the pregnant women who obliged and consented to partake throughout the research period.
- Maternal morbidity in 2005: estimates developed by WHO, UNICEF, UNFPA, and The World Bank, Geneva, World Health Organization. 2007. http://bit.ly/2rXGXhU
- Osunkalu VO, Taiwo IA, Makwe CC, Akinsola OJ, Quao RA. Methylenetetrahydrofolate reductase enzyme level and antioxidant activity in women with gestational hypertension and pre-eclampsia in lagos, Nigeria. J Obstet Gynaecol India. 2019; 69: 317-324. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/31391737
- Brown MA, Magee LA, Kenny LC, Karumanchi SA, McCarthy FP, Saito S, et al. Hypertensive disorders of pregnancy ISSHP classification, diagnosis, and management recommendations for international practice. Hypertension. 2018; 72: 24-45. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/29899139
- Akeju DO, Vidler M, Oladapo OT, Sawchuck D, Qureshi R, von Dadelszen P, et al. Community perceptions of pre-eclampsia and eclampsia in Ogun State, Nigeria: a qualitative study. Reprod Health. 2016; 13: 57. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/27357695
- Oloruntoba Ayodele Ekun, Oluwatumininu Mary Olawumi, Christian Chigozie Makwe, Nkeiruka Ogochukwu Ogidi. Biochemical assessment of renal and liver function among preeclamptic in Lagos metropolis. International Journal of Reproductive medicine. 2018. http://bit.ly/2M59J7d
- Gilles Guerrier, Bukola Oluyide, Maria Keramarou, Rebecca F Grais. Factors associated with severe preeclampsia and eclampsia in Jahun, Nigeria. Int J Womens Health. 2013; 5: 509-513. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/23983493/
- Khanum S, Naz N, Souza M. Prevention of pre-eclampsia and eclampsia. A systematic review. Open Journal of Nursing. 2018; 8: 26-44. http://bit.ly/2rXSsWC
- Santo Monte. Biochemical markers for prediction of pre-eclampsia: a review of the literature. J Prenatal Med. 2011; 5: 69-77. http://bit.ly/2YZSR6P
- Brandao AHF, Evangelista AA, Martins RMF, Leite HV, Cabral ACV. Prediction of early and late preeclampsia by flow-mediated dilation of the brachial artery. Radiol Bras. 2014; 47: 206-209. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/25741086
- Echeverria C, Eltit F, Santibanez JF, Gatica S, Cabello Verrugio C, Simon F. Endothelial dysfunction in pregnancy metabolic disorders. Biochim Biophys Acta Mol Basis Dis. 2019: 165414. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/30794867
- Griffin M, Seed PT, Duckworth S, North R, Myers J, Mackillop L, et al. Predicting delivery of a small for gestational age infant and adverse perinatal outcome in women with suspected pre-eclampsia. Ultrasound Obstet Gynecol. 2018; 51: 387-395. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/28401605
- Adamson Lee S. sFLT1 in preeclampsia: trophoblastic defense against a decidual VEGFA barrage. J Clin Invest. 2014; 124: 4690-4692. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/25329689/
- Adeosun OG, Charles Davies MA, Ogundahunsi, OA Ogunlewe J. Preliminary evaluation of hormonal and metabolic dysfunction: potential indicators of pre-eclampsia in African black women, southwest Nigeria. Sky Journal of Medicine and Medical Sciences. 2016; 4: 7-13.
- Sachidhanandam M, Singh SN, Salhan AK, Ray US. Evaluation of plasma hormone concentrations using enzyme immunoassay/enzyme-linked immunosorbent assay in healthy Indian men: effect of ethnicity. Indian J Clin Biochem. 2010; 25: 153-157. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/23105902/
- National High Blood Pressure Education Program. Report of the National High Blood Pressure Education program Working Group on high blood pressure in pregnancy. Am J Obstet Gynecol. 2000; 183: 119-122. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/10920346
- Rendon Macias ME, Villasis Keever MA. The research protocol V: The calculation of sample size. Rev Alerg Mex. 2017; 64: 220-227. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/28658730
- Kenneth L, Hall DR, Gebhardt S, Grove D. Late-onset preeclampsia is not an innocuous condition. Hypertens Pregnancy. 2010; 29; 3: 262 - 270. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/20670151
- Moshood Omotayo, Katherine Dickin, Rebecca Stolzfus. Perinatal mortality due to pre-eclampsia in Africa: a comprehensive and integrated approach is needed. Glob Health Sci Pract. 2016; 4: 350-351. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/27353628
- Adeosun OG, Charles Davies MA, Ogundahunsi OA, Ogunlewe J. Maternal and neonatal outcomes of pre-eclampsia in African black women, southwest Nigeria. Greener Journal of Medical Sciences. 2015; 5: 67-76. http://bit.ly/35uwJnE
- Dymara Konopka W, Laskowska M, Oleszczuk J. Preeclampsia - current management and future approach. Curr Pharm Biotechnol. 2018; 19: 786-796. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/30255751
- Ukah UV, De Silva DA, Payne B, Magee LA, Hutcheon JA, Brown H, et al. Prediction of adverse maternal outcomes from pre-eclampsia and other hypertensive disorders of pregnancy: A systematic review. Pregnancy Hypertens. 2018; 11: 115-123. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/29198742
- Shen M, Smith GN, Rodger M, White RR, Walker MC, Wen SW. Comparison of risk factors and outcomes of gestational hypertension and pre-eclampsia. PLOS One. 2017; 12: 0175914. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/28437461
- Yesmin F, Islam MS, Ferdoushi S, Faisal FM, Rehena Z, Afroza F, et al. Evaluation of serum ferritin concentration in mild and severe pre-eclamptic women. Mymensingh Med J. 2016; 25: 119-25. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26931260
- Kanika Mandi Choudhurya, Munmun Dasb, Sulekha Ghosh, Debasis Bhattacharyad, Tapan Kumar Ghoshe. Value of serum β-hCG in the pathogenesis of pre-eclampsia. J Clin Gynecol Obstet. 2012; 1: 71-75. http://bit.ly/35zbcua
- Çintesun E, Incesu Çintesun FN, Ezveci H, Akyurek F, Çelik C. Systemic inflammatory response markers in preeclampsia. J Lab Physicians. 2018; 10: 316-319. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/30078969/
- Feldt-Rasmussen U, Mathiesen ER. Endocrine disorders in pregnancy: physiological and hormonal aspects of pregnancy. Best Pract Res Clin Endocrinol Metab. 2011; 25: 875-884. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/22115163
- Fowden AL, Forhead AJ, Sferruzzi Perri AN, Burton GJ, Vaughan OR. Review: Endocrine regulation of placental phenotype. Placenta. 2015; 36: 50-59. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/25524059
- Praveen Sharma, Sunita Maheshwari, Shivani Barala. Correlation between second-trimester beta-human chorionic gonadotropin levels and pregnancy outcome in the high-risk group. International Journal of Reproduction, Contraception, Obstetrics and Gynecology. 2016; 2358-2361. http://bit.ly/2EqDY46
- Phipps EA, Thadhani R, Benzing T, Karumanchi SA. Pre-eclampsia: pathogenesis, novel diagnostics, and therapies. Nat Rev Nephrol. 2019; 15: 275-289. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/30792480
- Sharony R, Zipper O, Amichay K, Wiser A, Kidron D, Biron Shental T, et al. The magnitude of elevated maternal serum human chorionic gonadotropin and pregnancy complications. J Obstet Gynaecol. 2017; 37: 576-579. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/28604182
- Kaur G, Jain V, Mehta S, Himani S. Prediction of PIH by maternal serum beta HCG levels in the second trimester (13-20 weeks) of pregnancy. J Obstet Gynaecol India. 2012; 62: 32-34. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/23372286/
- Fisher SJ. Why is placentation abnormal in preeclampsia? Am J Obstet Gynecol. 2015; 213: 115-122. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26428489
- Ilekis JV, Tsilou E, Fisher S, Abrahams VM, Soares MJ, Cross JC, et al. Placental origins of adverse pregnancy outcomes: potential molecular targets. Am J Obstet Gynecol. 2016; 215: 1-46. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26972897
- Haavaldsen C, Fedorcsak P, Tanbo T, Eskild A. Maternal age and serum concentration of human chorionic gonadotropin in early pregnancy. Acta Obstet Gynecol Scand. 2014; 93: 1290-1294. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/25138917
- Alvarez Nava F, Soto M, Lanes R, Pons H, Morales Machin A, Bracho A. Elevated second-trimester maternal serum β-human chorionic gonadotropin and amniotic fluid alpha-fetoprotein as indicators of adverse obstetric outcomes in fetal Turner syndrome. J Obstet Gynaecol Res. 2015; 41: 1891-1898. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26369382
- Mallick MP, Ray S, Medhi R, Bisai S. Elevated serum β hCG and dyslipidemia in the second trimester as predictors of subsequent pregnancy induced hypertension. Bangladesh Med Res Counc Bull. 2014; 40: 97-101. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26402973
- Shahid A Mujawar, Vinayak W Patil, Rekha G Daver. Serum human chorionic gonadotropin as a biochemical marker of adverse pregnancy outcome in severe preeclampsia. American Journal of Biochemistry. 2018; 8: 13-17. http://bit.ly/2S2xzEk
- Settiyanan T, Wanapirak C, Sirichotiyakul S, Tongprasert F, Srisupundit K, Luewan S, et al. Association between isolated abnormal levels of maternal serum unconjugated estriol in the second trimester and adverse pregnancy outcome. J Matern Fetal Neonatal Med. 2016; 29: 2093-2097. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/27480207
- Singh S, Daga MK, Kumar A, Husain SA, Kar P. Role of oestrogen and its receptors in HEV-associated feto-maternal outcomes. Liver Int. 2019; 39: 633-639. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/29979823
- Goetzinger KR, Singla A, Gerkowicz S, Dicke JM, Gray DL, Odibo AO. Predicting the risk of pre-eclampsia between 11 and 13 weeks' gestation by combining maternal characteristics and serum analytes, PAPP-A, and free β-hCG. Prenat Diagn. 2010; 30: 1138-1142. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/20936638

Sign up for Article Alerts